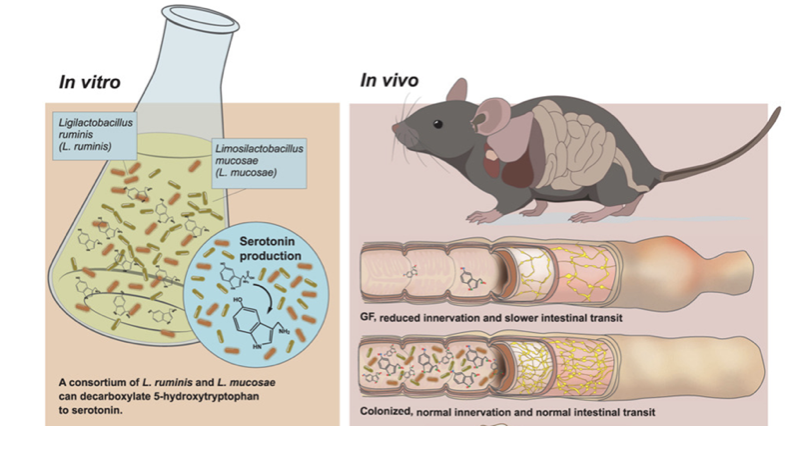
In the latest microbiome research, Moretti and colleagues uncover a surprising actor in gut physiology: specific human gut bacteria can synthesise bioactive serotonin and thereby influence intestinal nerve networks and motility. Until now, the bulk of serotonin in the gut (≈95 % of body’s serotonin is produced in the intestine) was believed to originate from host enterochromaffin cells via the Tph1‑mediated serotonin synthesis pathway. The study shows that gut microbes themselves can convert 5-hydroxytryptophan (5-HTP) into serotonin via decarboxylation, bypassing the classical hydroxylation of tryptophan step.
In experiments using germ-free and Tph1-deficient mice, microbial colonisation elevated faecal serotonin levels and increased colonic neuronal density and neurotransmitter-positive neurons — in effect, enhancing enteric nervous system (ENS) architecture and accelerating intestinal transit (i.e., motility) relative to controls.
On the microbial side, the authors isolated consortia from human stool that produced serotonin in culture, then traced the activity to a pairing of Limosilactobacillus mucosae and Ligilactobacillus ruminis — neither species alone in monoculture generated detectable serotonin in the tested conditions, suggesting a community synergy.
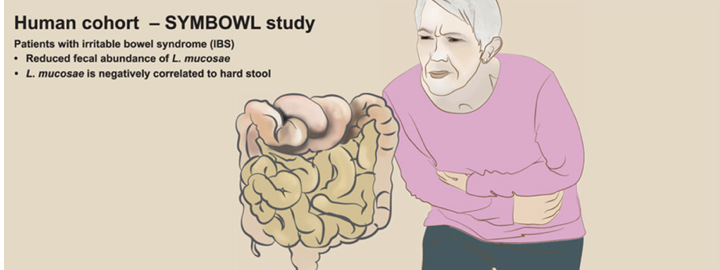
From an investment or therapeutic-development perspective, this opens a novel axis: microbial modulation of serotonin signalling, ENS development and gut motility. Conditions like Irritable Bowel Syndrome (IBS) — which feature dysregulated serotonin, altered microbial composition and impaired motility — may be approached via targeted microbial therapies or engineered probiotics to restore or boost microbial serotonin production. Indeed, the study notes lower abundance of L. mucosae in IBS patients.
Key caveats: this is pre-clinical work (mouse models, human isolates in vitro). Translation to humans remains to be validated: dose, colonisation, safety, inter-individual variability and regulatory hurdles are substantial. The mechanism is also not universal: only the defined consortium produced serotonin, and only under particular culture/host conditions.
In short, the paper reveals a previously hidden microbial pathway with potential to reshape gut-brain, gut-nerve and motility therapeutics — a promising frontier for microbiome-based investment and research pipelines.
Most important findings
- Human gut bacteria (specifically L. mucosae + L. ruminis) can convert 5-HTP into serotonin via decarboxylation.
- Microbial serotonin production was confirmed in vitro and in vivo (germ-free/Tph1-deficient mice) with increased faecal serotonin, enhanced colonic neuronal density and faster intestinal transit.
- Microbial serotonin production depends on a consortium/community effect (neither species alone produced serotonin under tested conditions).
- The gut microbiota represents a source of bioactive serotonin beyond host enterochromaffin cells.
- Lower abundance of L. mucosae in IBS patients suggests a potential link between microbial serotonin and functional bowel disorders.
- Opens novel therapeutic/investment opportunity: targeted microbial manipulation to modulate serotonin, ENS and motility in gut disorders.
- Translation remains early-stage: human applicability, colonisation dynamics, safety and regulatory pathways still untested.
Specific human gut bacteria can synthesise serotonin, enhance colonic neural networks and accelerate gut motility, offering a novel therapeutic axis for microbiome-based interventions in motility and gut-nerve disorders.
Data Metrics and Biological Insights
The study provides quantitative validation that microbial serotonin production is not only detectable but functionally relevant:
| Parameter | Germ-Free (GF) Control | Colonized with Limosilactobacillus mucosae + Ligilactobacillus ruminis (Ls) | Statistical significance |
| Fecal serotonin | Baseline ~0–50 ng/g | Elevated up to ~200–300 ng/g | p < 0.01 |
| Colonic neuronal density (Tuj1⁺ area) | Reduced baseline | ↑ ~40 % compared with GF | p < 0.05 |
| Serotonin-immunoreactive neurons (ENS) | Sparse | Significantly increased | p < 0.05 |
| Intestinal transit rate | Slowed (~5–6 h to expel marker) | Normalized to conventional-raised mouse levels (~3 h) | p < 0.01 |
| Host gene expression (Tph1, Mao) | Unchanged | No increase (proves microbial, not host origin) | ns |
Correlations were clear: fecal serotonin positively aligned with faster intestinal transit (Spearman r ≈ 0.6 in combined data, significant in females only).
Histological imaging confirmed enhanced neuronal networks within the myenteric plexus after colonization.
Clinical Correlation (IBS Cohort, n = 147 IBS vs 27 controls)
- L. mucosae abundance significantly lower in IBS (p < 0.01); L. ruminis unchanged.
- No statistically significant differences in circulating or fecal serotonin between IBS and controls—likely due to dynamic uptake and metabolism.
- Negative correlation between L. mucosae abundance and hard stool proportion (Spearman ρ ≈ –0.23; FDR < 0.05), linking bacterial loss to constipation-predominant phenotypes.
- Suggests that microbial serotonin deficiency may contribute to altered motility in IBS-C subtype.
Therapeutic and Investment Outlook
1. Commercial Viability
The study demonstrates a proof-of-concept for bio-engineered or precision probiotics targeting serotonin pathways. Live-biotherapeutic products (LBPs) based on L. mucosae and L. ruminis could address IBS-C, postoperative ileus, or neurogastrointestinal dysmotility. BioGaia AB, which co-funded and co-authored the study, already holds IP positions and a pending patent (as disclosed in Cell Reports).
2. Market Potential
The global functional GI disorder therapeutics market exceeds USD 12 billion (2025 est.), with >40 % of IBS drugs targeting serotonin signalling (5-HT3/5-HT4). A microbiome-based approach could offer non-systemic modulation with fewer side effects than receptor agonists/antagonists.
3. Investment Considerations
- IP Positioning: patent filed on serotonin-producing bacterial strains and decarboxylase enzymes.
- Regulatory Path: Live biotherapeutics face FDA/EMA LBP guidelines; preclinical toxicology and stability testing remain.
- Competitive Landscape: Seres Therapeutics, Vedanta Bio, and 4D Pharma have comparable microbial drug pipelines; none currently target microbial serotonin synthesis.
- Risk Factors: Translational gap (mouse → human), strain-specific colonization, potential horizontal gene transfer of decarboxylase enzyme, and scalability of anaerobic fermentation.
4. Research Implications
For academic researchers, the identified decarboxylase gene (pyridoxal-dependent, encoded by L. mucosae) provides a tractable molecular target. Future work will map environmental and nutritional cofactors (oxygen gradient, 5-HTP substrate availability) enabling microbial serotonin synthesis in vivo.
Quantified evidence now shows specific human gut bacteria generate serotonin, remodel intestinal nerve circuits, and normalize motility—laying biochemical and commercial groundwork for next-generation microbiome-based neuromodulators.
—
Pictures from University of Gothenburg