At the edge of a laboratory bench in Stavanger, a coin-sized battery sits in an air bath, looking inert, almost trivial. It does not move. It does not speak. Yet, as Jelena Popovic‑Neuber and her colleagues quietly insist, it contains a life, one that unfolds invisibly and unevenly across time. We measure batteries in cycles, in percentages, in ranges remaining on dashboards. But the true life of a battery, the one that matters, is happening elsewhere, at scales too small to see and too complex to narrate in any linear way.
To understand that hidden lifespan is to accept that a battery does not simply age. It transforms, negotiates with itself, resists, and eventually yields.
The thin layer that decides everything
Inside a lithium battery, where metal meets liquid, a boundary forms almost immediately. Researchers call it the solid electrolyte interphase, or SEI. The name has a bureaucratic flatness that conceals its significance. In reality, this layer governs almost everything that follows.
The SEI is a product of breakdown. When an electrode encounters an electrolyte, it reacts, decomposes molecules, and produces a thin film of organic and inorganic fragments. This layer is paradoxical. It protects the electrode by preventing further uncontrolled reactions, but it also obstructs the movement of ions and introduces resistance. It is, in Popovic‑Neuber’s formulation, both a protector and a problem.
If one were inclined toward metaphor, one might say that the battery builds its own scar tissue at birth.
What recent work shows, however, is that this scar is not static. A study in Communications Chemistry reveals that the SEI evolves over time into a structure that can be porous, layered, and partly liquid, partly solid, its morphology shaped by electrolyte chemistry, trace water content, and temperature. The layer is only nanometres thick, yet its internal architecture determines how easily ions can pass and therefore how efficiently the battery can charge, discharge, and endure.
We tend to think of ageing as gradual decline. But here ageing is architecture.
A system that ages in many directions at once
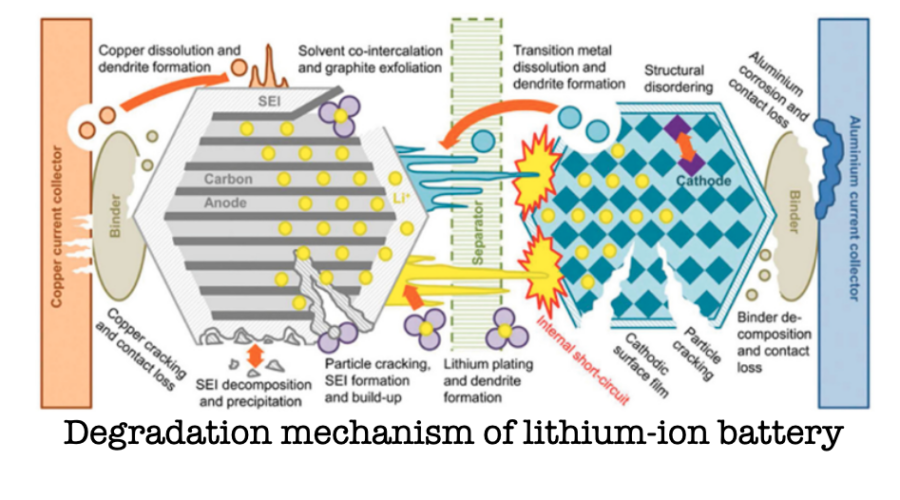
Even at a macroscopic level, the notion of battery ageing resists simplicity. Engineers like to define it in terms of capacity fade or increased internal resistance. But these are symptoms, not causes.
The degradation of lithium-ion batteries is not singular but plural, a set of overlapping processes influenced by chemistry, usage patterns, temperature, and time. Charge too quickly, discharge too deeply, expose the cell to heat or cold, and different pathways of decay accelerate.
Inside the battery, multiple failures unfold simultaneously. The electrolyte decomposes. Electrodes crack or swell. Lithium plates where it should not, sometimes forming dendrites that can puncture internal structures. Each process has its own tempo, and they do not necessarily align.
There is something unnerving in this multiplicity. We like systems that fail for one reason at a time, systems that can be diagnosed and repaired with clarity. Batteries, by contrast, fail as ecosystems do, through a web of interdependent stresses.
In one sense, the battery is less like a machine than a habitat.
Materials on the edge of possibility
At the centre of contemporary battery research lies a persistent ambition to go beyond the current limits of lithium-ion technology. The constraints are well known. Graphite anodes, for example, are reliable but limited in capacity. Silicon and alkali metals promise dramatically higher energy densities, but they introduce instability.
Silicon can store nearly ten times as much lithium as graphite, but its structure expands and contracts so violently during cycling that it fractures, forcing the SEI to repeatedly break and reform. Alkali metals such as sodium or potassium offer abundance and cost advantages, yet their electrochemical behaviour remains poorly understood, especially at the interface with liquid electrolytes.
The frontier, then, is not simply about discovering new materials. It is about managing instability. Increasing energy density inevitably brings the system closer to chemical extremes, where reactions are faster, interfaces more fragile, and failure modes more abrupt.
The paradox is that progress depends on flirting with breakdown.
Defects, imperfections, and the secret logic of performance
If one listens closely to the language of battery science, a curious theme emerges. Imperfection is not just tolerated; it is essential.
Defects within electrode materials, whether atomic vacancies, impurities, or structural dislocations, can either impede ion transport or facilitate it. Some defects enable conductivity. Others block it. The distinction is subtle and often only discernible through computational modelling and advanced spectroscopy.
What this suggests is a different philosophy of engineering. Instead of striving for ideal, flawless materials, researchers increasingly aim to design controlled imperfection. The goal is to cultivate defect landscapes that enhance performance without triggering instability.
In this sense, the battery becomes a negotiated object, its behaviour emerging from the balance between order and disorder.
The problem of knowing how long a battery will live
Despite decades of research, predicting battery lifespan remains a persistent challenge. Ageing is nonlinear. Cells that appear stable can degrade rapidly under certain conditions, while others endure longer than expected.
Machine learning approaches now attempt to make sense of this complexity by analysing real-world data, combining temperature, charge patterns, and usage histories to estimate remaining useful life. Yet even these models are constrained by the diversity of conditions and the scarcity of comprehensive datasets.
The difficulty is not merely technical. It is epistemological. We are trying to measure a system whose internal state cannot be fully observed without destroying it. We infer its condition indirectly, through voltage curves and impedance spectra, like physicians diagnosing an illness from symptoms alone.
There is an element of guesswork that cannot be eliminated.
The battery and the world it inhabits
The question of lifespan is not confined to the laboratory. It extends outward into the wider economy and environment.
Lithium-ion batteries depend on materials such as lithium, cobalt, and nickel, whose extraction carries significant environmental and geopolitical costs. Extending battery life is therefore not only a matter of efficiency but of ethics. Every additional cycle represents deferred extraction, reduced waste, and a smaller planetary footprint.
At the same time, batteries underpin the transition to renewable energy, from electric vehicles to grid storage systems. Their reliability shapes the feasibility of decarbonisation itself.
It is tempting to view the battery as a tool for solving external problems. Yet, as this research makes clear, the battery is itself a problem, intricate and unresolved.
Time, hidden and visible
There is a peculiar asymmetry in how we experience batteries. We notice them when they fail, when a device shuts down or a vehicle refuses to start. The rest of the time, they recede into the background, silently cycling through states of charge and discharge.
But inside the battery, time accumulates continuously. Each cycle leaves traces. Each chemical reaction alters the structure, however slightly. The SEI thickens. Defects emerge or migrate. Resistance grows.
The hidden lifespan is not a clock counting down but a history being written.
To understand that history is to accept that there may never be a single answer to the question of how long a battery will last. Instead, there are trajectories, shaped by choices of materials, patterns of use, and conditions of operation.
And perhaps that is the deeper lesson. The battery, often treated as a symbol of stored energy, is equally a symbol of stored time. Not time as we measure it, in hours or cycles, but time as it is lived by matter, through transformation, adaptation, and eventual decline.
We may never see that process directly. But in the quiet work of researchers, tracing reactions at the scale of atoms and interfaces, we come a little closer to understanding it.
References
Madani, S. S., Shabeer, Y., Allard, F., Fowler, M., Ziebert, C., Wang, Z., Panchal, S., Chaoui, H., Mekhilef, S., Dou, S. X., See, K., & Khalilpour, K. (2025). A comprehensive review on lithium-ion battery lifetime prediction and aging mechanism analysis. Batteries. [mdpi.com]
Popovic‑Neuber, J. (2026). How can we understand the hidden lifespan of batteries? University of Stavanger. [uis.no]
Grill, J., & Popovic‑Neuber, J. (2024). Long term porosity of solid electrolyte interphase on model silicon anodes with liquid battery electrolytes. Communications Chemistry. [colab.ws]
Rahman, T., & Alharbi, T. (2024). Exploring lithium-ion battery degradation: A concise review. Batteries. [mdpi.com]
Zhang, Y. (2024). Data-driven battery aging diagnostics and lifetime extension (Doctoral dissertation, Chalmers University of Technology). [research.chalmers.se]
University of Oslo et al. (2026). Revisiting the electrochemical response of alkali metals in contact with liquid battery electrolytes. Advanced Energy Materials. [mn.uio.no]
Photo from “A concise review. Batteries. [mdpi.com]“